How does Coptyzin work to control Psa in Kiwifruit
The unique chemistry of Coptyzin:
The chemistry behind Coptyzin is quite different to that of salt based coppers.
The TEPA chelating agent is a long chain of polyamines and polyethyleneimines which are able to form very stable chelates. The high stability constant of 22.4 (logarithmic value) allows the CuTEPA to release the copper ions gradually over a period of time.
The structure of the TEPA differs to that of other copper chelating agents such as DTPA, EDTA and HEEDTA. The TEPA slows the rate of absorption into the leaf ( which is mainly via the stomata and cuticles ) thereby reducing the risk of phytotoxicity.
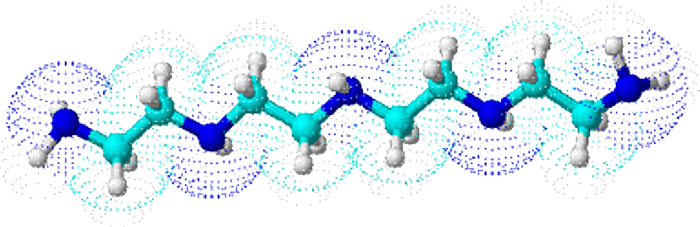
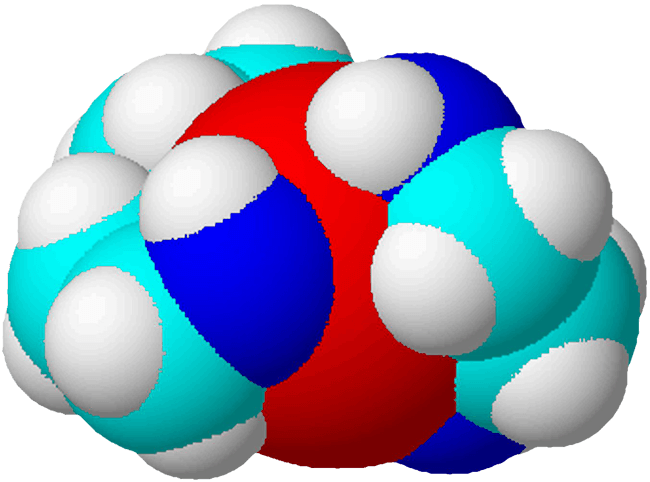
Copper TEPA Chelate
The CuTEPA chelate is not only highly soluble, but it is also positively charged.
Leaves have negative charges on the outside (they have a cation exchange capacity CEC) and therefore they will retain cations and positively charged molecules such as the CuTEPA chelate in Coptyzin, thereby increasing the copper concentration on the outside of the leaves.
Coptyzin covers the leaves with copper a lot more evenly. It creates a very fine film of copper, (almost up to molecular level) affording protection using a much smaller dosage compared to other copper sprays.
Copper Content of Coptyzin compared with commonly used competitor products
| Cuprous Oxide Nordox |
TEPA Cu Coptyzin |
Copper Hydroxide Kocide Opti |
|
|---|---|---|---|
| Budbreak to flowering period typical dosage as per Zespri CPS |
37.5G/100L | 95G/L | 70G/100L |
| Copper content - grams @ 1000l water per ha | 281.25 | 142.5 | 210 |
| Difference in grams | 138.75 | 67.5 |
|---|---|---|
| Difference ( times ) | 1.97 | 1.47 |